The Tale of the Tail: Clues from Mice
As mice age, their tails often become stiff and kinked. However, in the lab of molecular biologist Shin-Ichiro Imai at Washington University School of Medicine in St. Louis, certain genetically modified mice maintained limber and nearly straight tails even as they aged. These mice displayed other signs of defying aging, such as being more robust and spending more time on exercise wheels compared to their normal counterparts. Most notably, these genetically altered mice lived about 7% longer, gaining an extra 58 days of life, as reported by Imai and colleagues in Cell Metabolism.
The genetic modification enhanced a key communication signal from the brain to the body. Specifically, a group of neurons in the hypothalamus, a control center deep in the brain, remained active as the animals aged. Imai’s team found that these neurons send signals to the animal’s fat stores via the sympathetic nervous system, prompting the fat to burn lipids and secrete a molecule called NAMPT. This molecule helps prevent aging-related damage in other parts of the body, including the hypothalamus itself.
The Breakdown of Communication
As mice age, the neurons that send the initial signal typically falter, Imai’s group discovered. This leads to a lack of proper stimulation of fat stores, causing the animals to accumulate fat, become sedentary, and produce less NAMPT. Essentially, this communication breakdown accelerates physical decline. Counteracting it appeared to keep the mice spry.
Imai and his team are among a growing number of scientists exploring how communication between organs and tissues might combat aging. Understanding this inter-organ communication is crucial for comprehending the aging process, according to geneticist Norbert Perrimon of Harvard Medical School. The National Institute on Aging has even begun requesting grant proposals to investigate these connections.
Long-Distance Signaling: Beyond Hormones
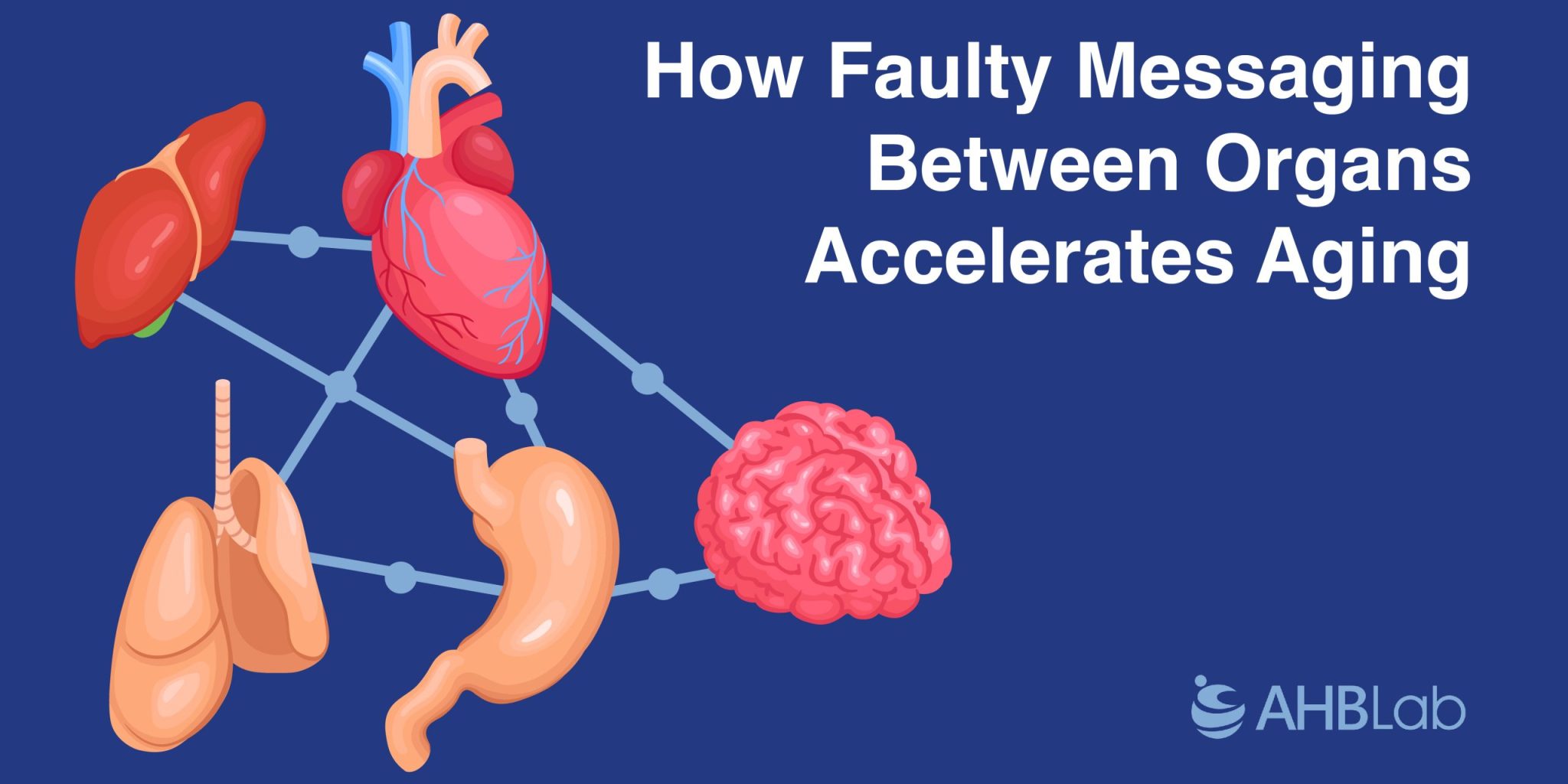
Over the past couple of decades, researchers have found that long-distance signaling involves more than just the well-known hormones from glands like the pancreas, thyroid, testes, and ovaries. Most organs and tissues now appear to bombard each other with a variety of molecular messages. For instance, fat dispatches more than 100 different signaling molecules, and muscles send out over 600. Even bones, once thought to be silent, communicate with muscles, the brain, and other organs.
The nervous system is also part of this expanding signaling network. Researchers are redefining anatomy with newfound connections in the nervous system, according to neuroscientist Ana Domingos of the University of Oxford. For example, pancreatitis, an inflammation of the pancreas, can send inflammatory signals to the lungs, making the condition fatal half of the time.
Mapping the Body’s Communication Network
Scientists are just beginning to catalog the body’s molecular chatter. Blood is rich with molecules that could be inter-organ messages, influencing physiology or aging. However, identifying these signaling molecules and their origins is challenging, says molecular biologist Ilia Droujinine of Scripps Research.
In 2021, Droujinine, Perrimon, and colleagues developed a technique to identify potential inter-organ signals. They genetically modified an organ within an animal to attach a marker, biotin, to proteins due to be secreted. These tagged proteins carry a badge of their origin as they travel through the body. Using this method in fruit flies, researchers found proteins that traveled from muscles to heads and from the fat body to legs. A follow-up study in 2022 confirmed similar results in mice.
The Brain’s Chat with Other Organs
A clearer picture is emerging of how the brain communicates with distant organs. Early anatomists mapped the nervous system through dissection. Now, better microscopes, upgraded markers for labeling neurons, and new techniques that make animals’ bodies transparent have revealed new connections.
For instance, evidence suggested that sympathetic nerves connect to white adipose tissue (fat), but it wasn’t confirmed in living animals until 2015. Domingos and colleagues found that nerves and fat cells make contact, with nerve fibers appearing to grip individual fat cells. Stimulating these neurons prompted fat cells to break down lipids, showing that the sympathetic nervous system plays a significant role in metabolism.
Aging and Communication Decline
As mice age, the brain-to-body link weakens. Imai’s team showed that as neurons in the hypothalamus become less active, sympathetic nerves retract from fat tissue, leading to less NAMPT production. This creates a cycle of dysfunction. The decline could be triggered by age-related inflammation in the hypothalamus or white adipose tissue.
The heart also loses nerve connections with age, as cardiovascular biologist Stefanie Dimmeler of Goethe University Frankfurt and her team revealed in 2023. They found that nerve fibers disappeared from the left ventricle of the heart in older mice, reducing heart rate variability and potentially predicting early death. Senescent cells, which accumulate with age, trigger this nerve retreat.
Inter-Organ Communication and Aging
Altered inter-organ communication can promote physical decline and speed aging. For example, geneticist Hua Bai of Iowa State University found that older fruit flies’ equivalent of the liver releases more cytokines, which cause the heart to beat abnormally. Similarly, the nematode Caenorhabditis elegans shows that mitochondrial damage can trigger a protective unfolded protein response in distant tissues, boosting longevity.
If these communication channels in animals are as powerful in humans, they could inspire new treatments for diseases and even counteract aging. For instance, new treatments for neurodegeneration could target an easier-to-access organ to pass benefits to the brain via inter-organ messages.
Promising Inter-Organ Signals
Of the thousands of inter-organ messages, some stand out for their therapeutic potential. Osteocalcin, a hormone from bones, induces metabolic benefits, such as stimulating the liver to release insulin and spurring muscles to absorb glucose. It also aids cognition. Geneticist Gerard Karsenty’s team is studying whether mice with extra osteocalcin live longer.
Imai’s discoveries about the hypothalamus and NAMPT could also lead to new treatments. NAMPT is packaged in extracellular vesicles (EVs) released from fat. Infusions of NAMPT-filled EVs from young mice allowed older rodents to live 10% longer. Imai envisions collecting and freezing people’s EVs for future infusions to slow aging.
Potential Therapies and Future Research
If the inter-organ communication pathways discovered in animals prove to be similarly influential in humans, they could revolutionize treatments for age-related diseases and overall longevity. Researchers are optimistic that the clues they have uncovered might pave the way for innovative therapies targeting these communication channels.
For example, Gerard Karsenty’s team at Columbia University is focusing on osteocalcin, a hormone produced by bones, which has been shown to offer multiple metabolic benefits, including enhancing insulin production by the liver and improving muscle glucose absorption during exercise. Intriguingly, it also appears to support cognitive functions. The potential of osteocalcin to counteract aging-related issues has led to studies where mice are genetically engineered to produce higher levels of this protein to assess if it can indeed extend lifespan. Though genetic engineering might not be practical for humans, Karsenty’s team is also developing a molecule to boost osteocalcin levels in mice, with hopes for human trials if proven safe and effective.
Shin-Ichiro Imai’s research on the hypothalamus and NAMPT offers another promising avenue. NAMPT is crucial for maintaining cellular health and is released from fat cells within extracellular vesicles (EVs). Imai’s studies demonstrated that older mice infused with NAMPT-filled EVs from younger mice lived 10% longer. This concept could potentially be applied to humans by collecting and storing a person’s EVs when they are younger for use later in life, essentially offering a rejuvenating boost from their own youthful cells.
The Importance of Understanding Inter-Organ Communication
Deciphering the complex language of inter-organ communication is key to unlocking new ways to promote healthy aging and treat age-related diseases. As researchers map these pathways and understand the specific messages organs send to each other, they can develop targeted therapies to enhance these communications, thereby supporting the body’s natural processes to maintain health and vitality.
While much of this research is still in its early stages, the potential implications are profound. If scientists can identify and manipulate the critical signals that maintain organ health and inter-organ communication, they could slow down or even reverse some of the detrimental effects of aging. This could lead to breakthroughs not only in extending lifespan but also in improving the quality of life in our later years.
The Future of Anti Aging Research
The ongoing research into inter-organ communication represents a new frontier in the field of aging and longevity. Scientists like Imai, Karsenty, and their colleagues are pioneering this field, uncovering how organs talk to each other and how these conversations affect overall health. As the body’s molecular messages are cataloged and understood, the next steps will involve developing practical interventions to enhance these communications.
Potential treatments could range from new medications that mimic the beneficial effects of hormones like osteocalcin and NAMPT, to lifestyle changes and dietary supplements designed to boost the body’s natural production of these molecules. Furthermore, the ability to harness and store the beneficial extracellular vesicles from a person’s younger years could become a cutting-edge approach to personalized anti-aging treatments.
Conclusion
The study of inter-organ communication is revealing just how interconnected and communicative our organs are. As researchers delve deeper into these complex networks, the potential to influence aging and promote longevity becomes more tangible. From genetically modified mice with youthful traits to potential human therapies that boost beneficial molecular signals, the future of anti-aging science looks promising.
Understanding and enhancing the body’s natural communication channels could unlock new ways to maintain health and vitality well into old age, transforming our approach to aging and significantly improving the quality of life for future generations. As this research progresses, it holds the promise of not just longer lives, but healthier, more vibrant ones, where the physical decline associated with aging becomes a thing of the past.
At AHB Lab, we’re not just leaders in peptide synthesis; we’re at the forefront of pioneering biotechnology exploration. Our commitment extends beyond peptide production to uncovering the molecular mysteries that drive groundbreaking health solutions. By aligning with the latest scientific research, including studies on inter-organ communication and aging, AHB Lab is dedicated to advancing our understanding of peptides and their potential in revolutionary biotech applications. Join us as we forge new paths in science, contributing to the future of biotechnology with our unwavering commitment to excellence and innovation. As a peptide manufacturer, we report the latest news and advancements to keep you informed and inspired.