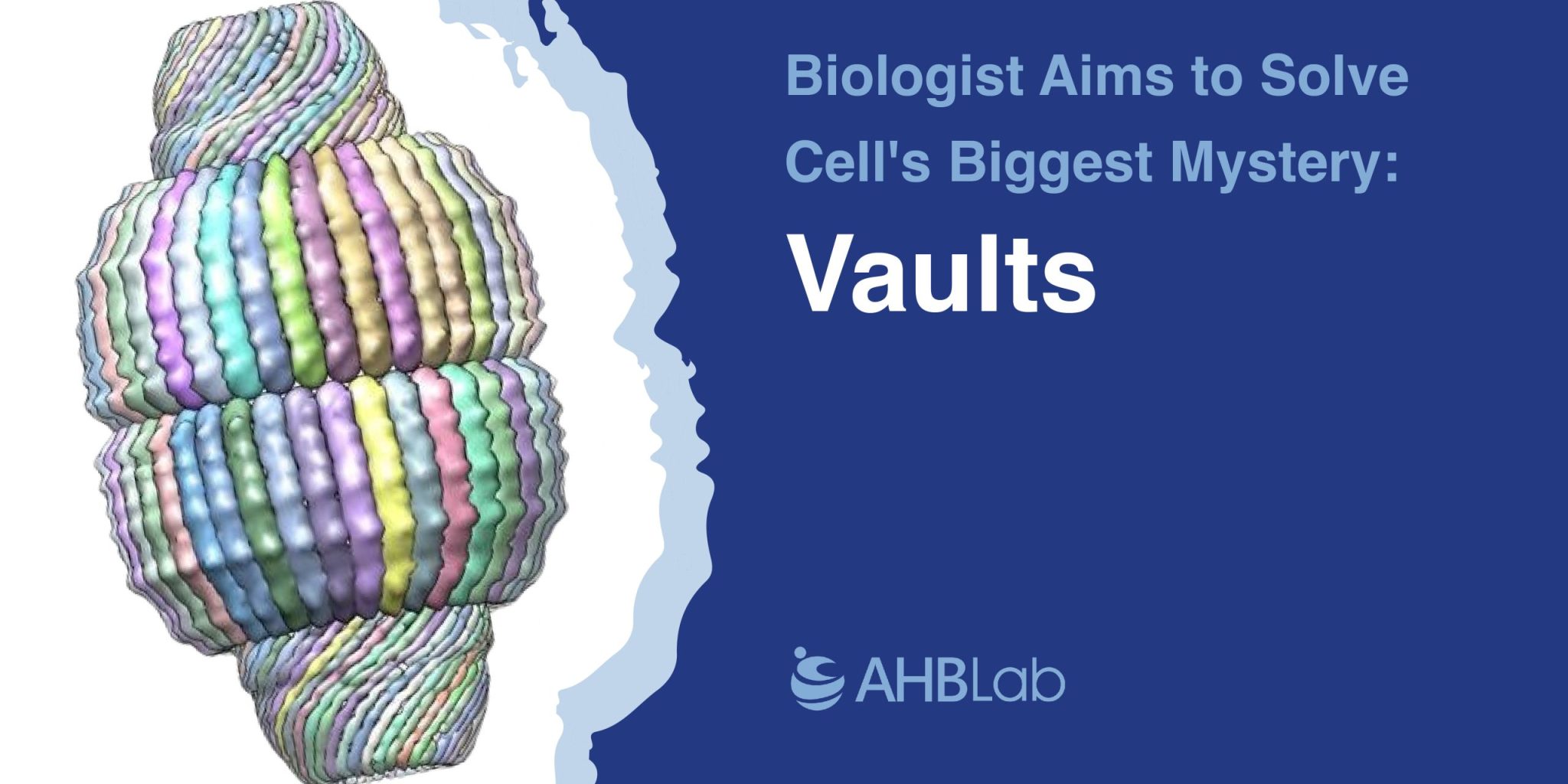
The Discovery of Vaults
Leonard Rome switches off the overhead light in a small room, leaving it illuminated only by a computer monitor and the fluorescent screen at the base of a towering electron microscope. Qing Lou, a Ph.D. student at the University of California, Los Angeles (UCLA), points to some ovoid smudges within the circular green glow of the microscope display. With a twist of a dial and a click of a mouse, she brings the shadows into focus and snaps a picture. Dozens, maybe hundreds, of barrel-shaped particles suddenly fill the computer monitor.
“There they are,” Rome says, like a proud father showing off his children.
These are vaults, enigmatic cellular structures that he and his then-postdoc Nancy Kedersha discovered back in 1986. Vaults, as he and others have shown, are the most massive particles made naturally by human cells and among the most abundant. Most of our cells have roughly 10,000 of these structures, with the number rising to perhaps 100,000 in certain immune cells. Their abundance—and the resources cells must pour into making them—suggests vaults have some essential function. But despite decades of work by Rome and other “vaulters,” their purpose remains unknown.
The Mystery of Vaults
Over the decades, various hypotheses have been proposed, including that vaults help ferry things around inside cells or clear toxins. Yet, one by one, promising ideas were dismissed or lost momentum as supporting evidence failed to materialize. Initially enthusiastic about Rome and Kedersha’s discovery, the National Institutes of Health (NIH) lost interest in funding basic research on vaults as the years wore on without answers.
Yet, Rome’s fascination with vaults hasn’t faded, even as other researchers—including Kedersha—moved on. Now, with help from other funders and labs, he has turned from basic research on vaults to studies of how they might be exploited in medicine and other fields, as nanoscale vessels for delivering therapies and more.
Vaults in Medicine
The ones in the microscope on this day were produced in genetically modified yeast and loaded with an immune signaling molecule called CCL21 that has shown tumor-fighting potential. Vault Pharma, a company co-founded by Rome that works out of an incubator space at UCLA, hopes to start a clinical trial in late-stage cancer patients as soon as this year. It would mark the first time synthetic vaults have been injected into humans and perhaps the beginning of a new turn in the spotlight for these mysterious organelles.
Rome’s Journey and Challenges
Rome is now 75, with three grown sons. His hair has turned white, and his impressive mustache is long gone. He retired in 2020 but is now officially back, unpaid, at UCLA with one lab bench, a postdoc, and some undergraduates under his wing. From a small office at UCLA’s California NanoSystems Institute (CNSI), which he helped design and, for a period, directed, he spends his days consulting on vault research with other UCLA labs and the small Vault Pharma team.
In one audacious effort, he and a lab in St. Louis have stuffed viruses into vaults in a bid to solve a major problem in gene therapy. He’s aided by his infectious enthusiasm. UCLA environmental engineer Shaily Mahendra years ago became a vault convert, loading the synthetic ones with enzymes designed to break down groundwater contaminants. She and Rome now have backing from foundations and federal agencies to see whether the encapsulation helps the enzymes work better and last longer when dispersed across land or in water.
The Origins of Vaults
Rome debated majoring in art or chemistry as an undergrad when his art teacher gently suggested the latter was a more practical option. He later switched to biochemistry, which at the time involved a lot of cell biology. His first lab at UCLA focused in part on a cellular structure: lipid spheres known as clathrin-coated vesicles. They transport enzymes to organelles called lysosomes to help break down other molecules.
When Kedersha joined Rome’s group in 1983, she was fresh out of a doctoral program at Rutgers University but had previously worked for years as a lab tech. The two were nearly the same age and quickly developed a rapport. Rome assigned her the task of comparing the vesicles entering lysosomes with the ones exiting. When she used a gel to separate different kinds of vesicles based on their electrical charge, she unexpectedly saw an additional band, indicating structures of another kind. Unable to identify them, she tried flooding her cell preparations with a heavy metal stain before looking at them with an electron microscope.
Naming the Vaults
Like islands in an ocean, white ovoid outlines appeared amid the background puddle of stain—vaults. The stain was rolling off the structures, revealing their shape. Follow-up experiments revealed that more traditional stains for the lipid membranes common to many cell components did not bind to vaults’ protein-only shells, one reason they had gone undiscovered for so long.
The lab held a competition to name the unidentified cellular objects, which were shaped like tiny U.S. footballs. Some suggested grenades or raspberries. Romesomes was a popular choice, which Rome quickly countered with Kedershacules. But when the postdoc remarked that she thought the objects’ outline resembled the vaulted ceilings in cathedrals, the name stuck.
The Continuing Mystery
Rome’s lab and a few others have built up a more detailed picture of vaults over the years. About 10 million times smaller than a football, a vault is still large for a cell—about three times the mass of the much better-known ribosomes that translate RNAs into proteins. Each is made up of 78 copies of the elongated major vault protein (MVP), aligned somewhat like staves in a barrel. Inside are clumps of two other proteins and the short vault RNA (vRNA).
The genes for these vault components are found in diverse eukaryotic organisms—those that pack their DNA in the nucleus and share other cellular features—with notable exceptions that include insects, plants, and fungi. Bacteria also seem to lack them. A 2013 study constructed a family tree of all the organisms known to have vaults and concluded they date back to a hypothetical last common eukaryotic ancestor billions of years ago. Over evolutionary time, some lineages evidently lost them.
Based on their barrel shape, Rome initially wondered whether vaults pick up and released cargo within the cell, perhaps plying routes from the nucleus to other locations. Vaults seemed to gather around gateways known as nuclear pore complexes and might fit their opening.
Vaults in Cancer Research
In 1996, European cancer researchers investigating a protein that’s unusually abundant in drug-resistant cancer cells cloned the gene and discovered that it resembled the gene for MVP in rats. Working with Valerie Kickhoefer in Rome’s lab, the group then found that the drug-resistant cancer cells generated many more vaults than nonresistant ones, suggesting the structures might sequester or expel chemotherapies. But to investigators’ frustration, stopping the production of vaults, or MVP, didn’t make the cells more susceptible to drugs.
Underscoring the mystery of vaults, in 2002 a Dutch team disabled the gene for MVP in a line of mice. The rodents lacked vaults yet developed normally, seemed to be healthy, reproduced, and lived as long as regular mice. Subsequent knockouts of the two other vault protein genes also left mice unscathed. And Vidigal’s team at NIH recently disabled the mouse vRNA gene—only to find that those rodents, too, display no major changes.
The Future of Vault Research
Despite these puzzling findings, Rome’s lab continues to explore the potential of vaults. They are now looking into using vaults to deliver gene therapies and vaccines. Vault Pharma, though facing funding challenges, is on the verge of a pivotal clinical trial for CCL21 cancer therapy.
Rome remains optimistic. He launched a series of online videos earlier this year to inspire a new generation of vaulters. On “The Vault Guy,” his YouTube channel, Rome mixes jokes and cartoonish animations with serious discussions of scientific methods, funding, and publishing. With barely more than 100 subscribers to date, the series isn’t exactly going viral. But some viewer just might be the person who finally solves the great mystery.
“The optimist in me is that before I die, someone will figure out what vaults do,” Rome says.
About AHB Lab
At AHB Lab, we’re not just leaders in peptide synthesis; we’re at the heart of pioneering biotechnology exploration. Our focus extends beyond mastering peptide production to embody a vision that drives innovation across the biotech landscape. We are committed to exploring the depths of peptide structure and function, uncovering the molecular mysteries that hold the key to groundbreaking health solutions. By aligning with the latest in scientific research and technological advancements, AHB Lab is dedicated to spearheading developments that enhance our understanding of peptides and pave the way for revolutionary biotech applications. Just as Leonard Rome’s relentless pursuit of the mysteries of cellular vaults is opening new doors in cancer therapy, AHB Lab’s commitment to excellence and innovation is shaping the future of biotechnology. Join our quest as we forge new paths in science and contribute to the evolving landscape of health and medicine.
Cover Image:
“Vault PDB structure” by H. Tanaka, K. Kato, et al., used under CC BY-SA 3.0.