On March 24th, 2023, AHB Lab received the approval of INCI for ICPPX® Peptide from the US INCI committee.
INCI stands for International Nomenclature of Cosmetic Ingredients.It is a standardized system of names for ingredients used in cosmetic and personal care products. The INCI system was established to promote consistency in labeling and to facilitate communication between companies and consumers. The approval of ICPPX®’s INCI from the US INCI committee is a significant achievement for AHB Lab. It is a testament to the quality and innovation of our products.
Check here to learn more about INCI.

Importance of INCI Approval of ICPPX® Peptide
INCI approval is an essential requirement for cosmetic and personal care products. It serves as a universal language for consumers and manufacturers alike. By using standardized names for ingredients, companies can ensure consistency in labeling and avoid confusion among consumers. INCI also helps consumers make informed decisions by providing information about the ingredients used in products.
INCI approval is crucial for AHB Lab to effectively market and expand our customer base. The approval of ICPPX®’s INCI allows us to promote the benefits of ICPPX® Peptide and reach a wider audience. INCI approval also enhances our credibility as a company and demonstrates our commitment to quality and innovation.
AHB Lab’s Breakthrough Peptide Receives US INCI Approval
The approval of ICPPX®’s INCI from the US INCI committee is a significant achievement for AHB Lab. It reflects our commitment to innovation and quality. As well as our dedication to providing our customers with the best possible products. The approval of ICPPX’s INCI is also a milestone in the development of ICPPX® Peptide. Because it further validates the safety and efficacy of our product.
AHB Lab produces and refines ICPPX® Peptide through a fermentation process, utilizing endotoxin-free E. coli or edible pharmaceutical-grade yeast. Protease hydrolysis technology is used to hydrolyze the specific ICPPX® peptides from the protein, followed by purification and refinement to create a high-precision and high-concentration product. With the approval of ICPPX®’s INCI, we can now promote the benefits of ICPPX® Peptide more effectively and reach a wider audience.
Introduction of ICPPX®
Check here to learn more about ICPPX® Peptide.
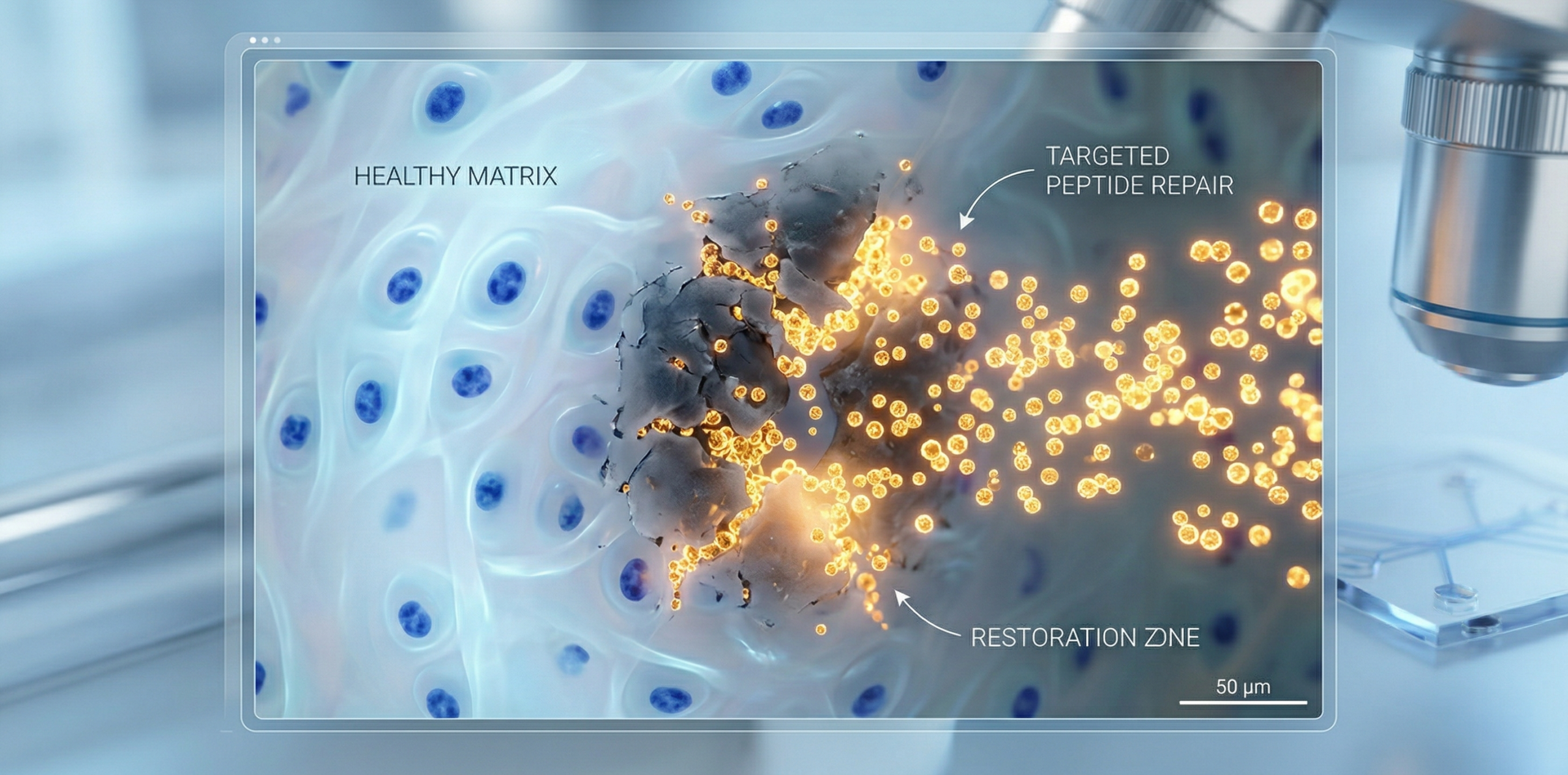
ICPPX® Peptide is a revolutionary therapy developed by AHB-Lab’s patented Synthetic Biology Peptidomimetics Platform (SBPP). ICPPX® is an innovative hexapeptide and pentapeptide targeting degenerative arthritis, tendinopathy, myofascial pain, sprain, and muscle ache, eliminating stasis and subdue swelling. It may stimulate and activate joint and tendon fibroblasts to synthesize collagen I, collagen II, hyaluronic acid, and fibronectin through three mechanisms, including fibroblast, chondrocyte, and TGF-β growth factor secretion. Secondly, ICPPX® Peptide may also have simultaneous anti-inflammatory and pain-relieving effects by decreasing IL-1β and TNFα concentrations in inflamed degenerative arthritis, tendon fasciitis, and ligamentitis tissue.
Potential Applications of ICPPX® Peptide
ICPPX® Peptide has the potential to revolutionize the treatment of various health conditions, including joint and tendon health. Its ability to stimulate collagen synthesis and reduce inflammation makes it an ideal therapy for these conditions. We primarily target arthritis and sports-related conditions with ICPPX® Peptide. Furthermore, ICPPX® may offer benefits such as anti-inflammation, pain relief, tissue repair, and reducing swelling. Specifically, it may activate joint and tendon fibroblasts to synthesize collagen I, collagen II, hyaluronic acid, and fibronectin. Moreover, ICPPX® may have simultaneous anti-inflammatory and pain-relieving effects by decreasing IL-1β and TNFα concentrations in inflamed degenerative arthritis, tendon fasciitis, and ligamentitis tissue. Additionally, for athletes and individuals suffering from sports-related conditions, ICPPX® Peptide may also assist with tendinopathy, myofascial pain, sprain, and muscle ache by eliminating stasis and subduing swelling.
Conclusion
In conclusion, the approval of ICPPX®’s INCI is a significant achievement for AHB Lab. And it serves as a testament to the quality and innovation of our products. We are excited to bring ICPPX®, a breakthrough therapy with the potential to improve the lives of many, to the market. With the approval of ICPPX®’s INCI, we can now effectively promote its benefits to a wider audience. Furthermore, broadening our customer reach will enable us to advance our goal of promoting healthier and happier lifestyles for individuals.
As we continue our research and development efforts, we remain dedicated to bringing more innovative and high-quality products to the market. Our ultimate goal is to make a positive impact on people’s lives. And we believe that ICPPX® Peptide is just the beginning of our journey towards achieving this goal.





One Response